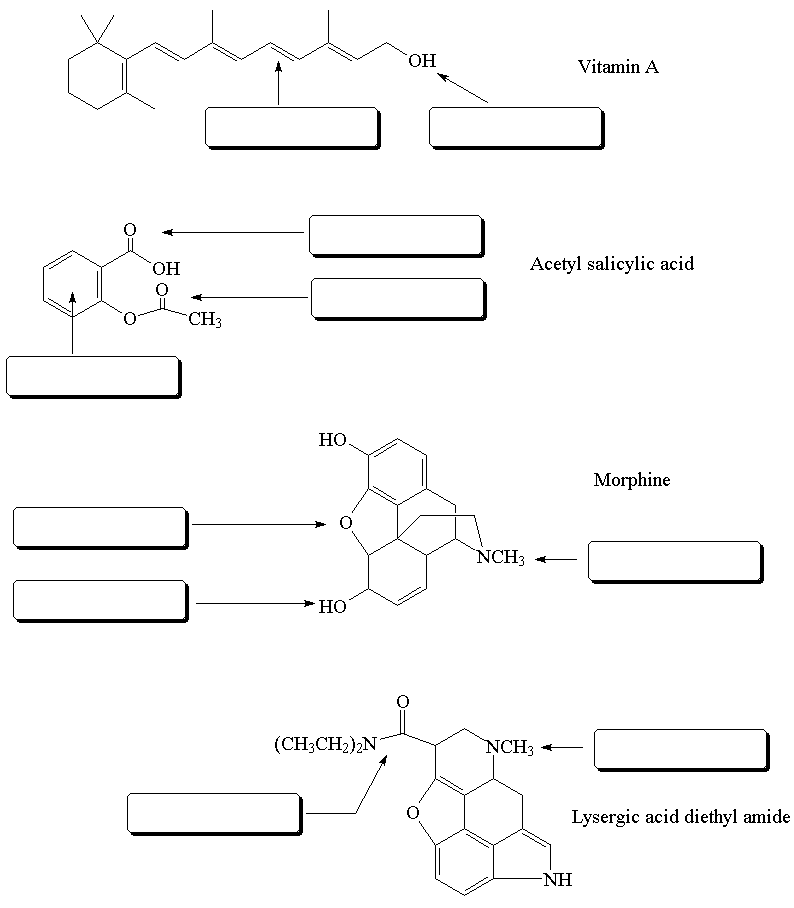
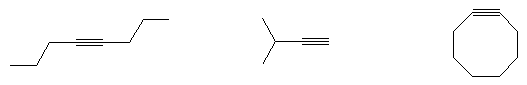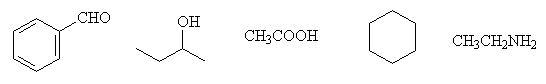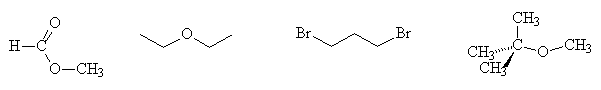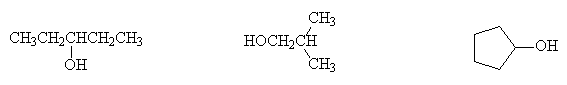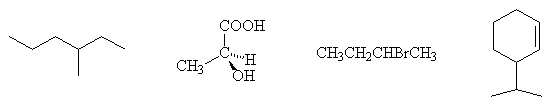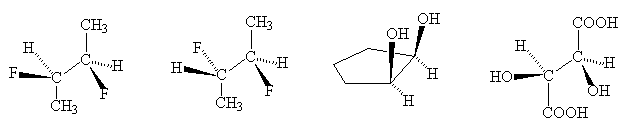QUESTIONS
These are the questions that appear in the course notes and are by no means exhaustive! Use pen and paper to work out the answers and then check them against those on the answer page accessed by clicking "answers" at the end of each section. If you are not sure of the answer go back to the appropriate page of the course notes.
INTRODUCTION
- Name the functional groups in the following molecules:
Go to answers
Return to Introduction
Go to Index
ALKANES
- Draw all the constitutional isomers of C5H12 and C6H14.
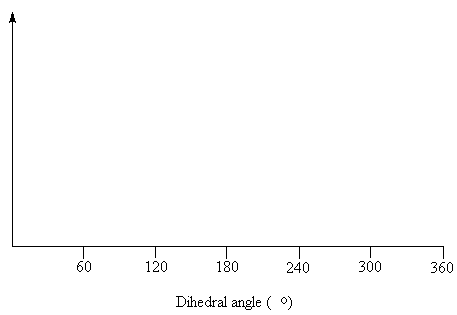
- Draw the Newman projections of the conformers representing the maximum and minimum points on the energy profile. Draw the energy profile of a butane molecule as the C2-C3 bond is rotated through 360° .

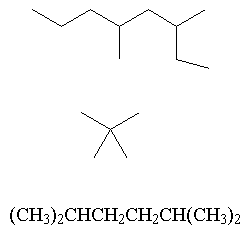
- Name the following compounds:
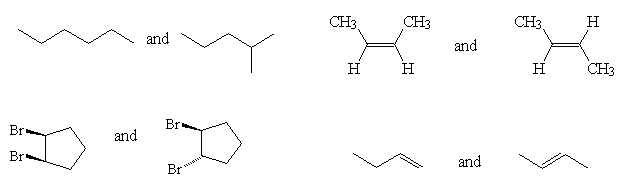
- Classify the following pairs of molecules as Constitutional isomers, Conformational isomers or Diastereoisomers.
Go to answers
Return to Alkanes
Go to Index
ALKENES
Give the names of the following molecules:
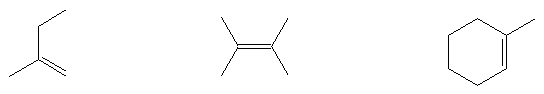
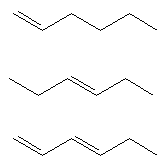
- Name these molecules (include Z and E where appropriate)

- Draw the curly arrow in the following reaction:
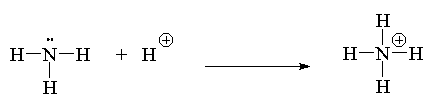
- Use arrow notation to illustrate the mechanism of the reaction of Br2 with propene. Label each species electrophile or nucleophile.
Go to answers
Return to Alkenes
Go to Index
ALKYNES
- Name the following compounds
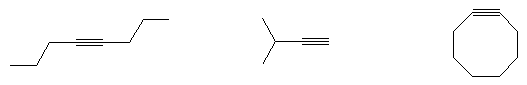
- Predict the products in the following reactions:
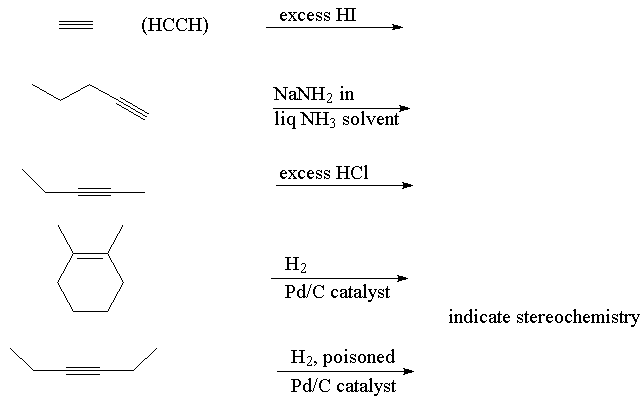
Go to answers
Return to Alkynes
Go to Index
AROMATIC COMPOUNDS
- Fill in the missing reagents or products in the following reactions:
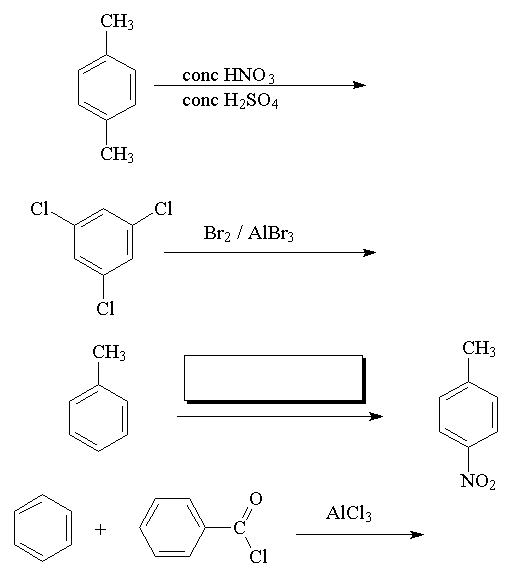
Go to answers
Return to Aromatic Compounds
Go to Index
SPECTROSCOPY
What is the value of m/z of the molecular ions (integer values) of the following compounds?

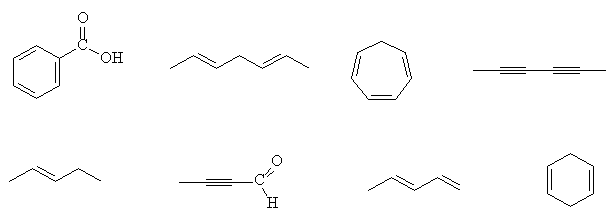
- Which of the following compounds strongly absorb electronic radiation in the ultraviolet-visible region of the spectrum ?
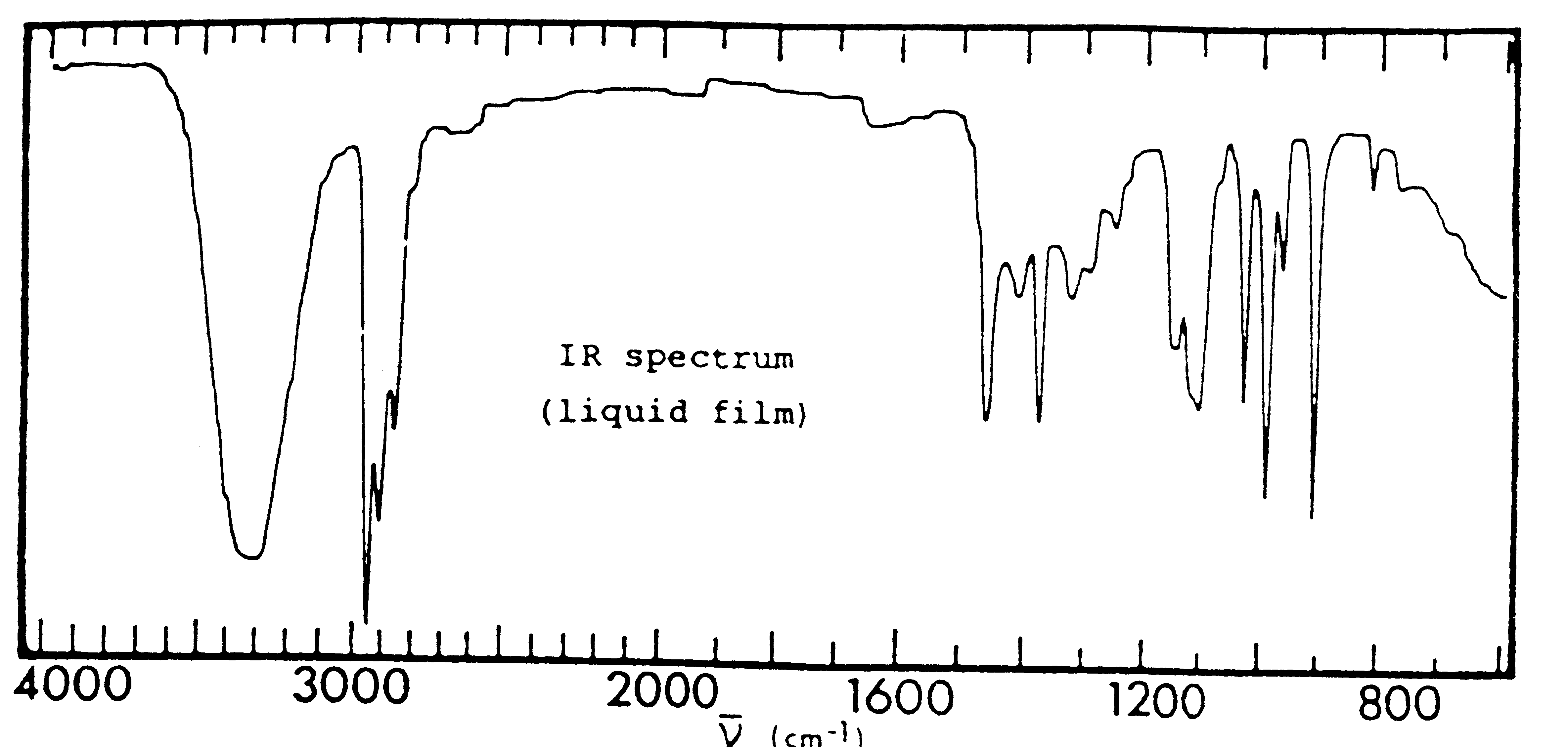
- Which of the compounds: CH3CH2COCH3, CH3CH2OCH3, CH3CH2CH2OH, is the following infrared spectrum representative of?
- Indicate which of the following compounds absorb around 1700 cm-1 and around 3500 cm-1 in the infrared region.
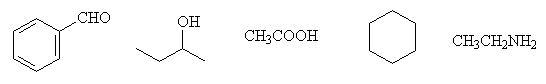
- A compound with molecular formula C3H8O has a strong absorption at 3600 cm-1 in the infrared spectrum. Give a reasonable condensed structural formula for any compound whose structure is consistent with this data.
- How many signals would be expected in the 1H NMR spectrum of each of the following compounds?
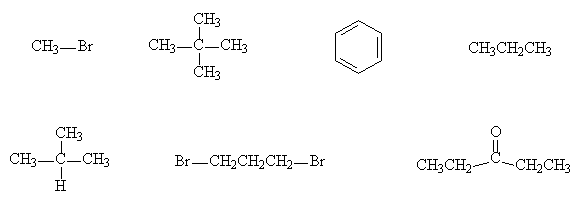
- Give the ratio of signal sizes observed in the 1H NMR spectrum of each of the following molecules:
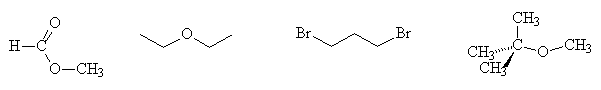
- Give the number of signals, their relative intensities and the multiplicities in the 1H NMR spectra of each of the following compounds:
Go to answers
Return to Spectroscopy
Go to Index
ALCOHOLS
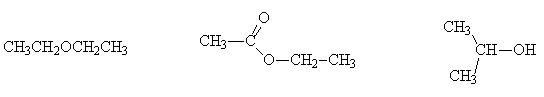
Name and classify the following alcohols as primary, secondary or tertiary
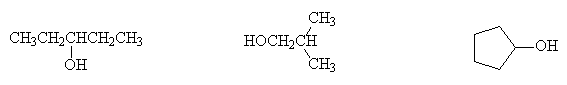
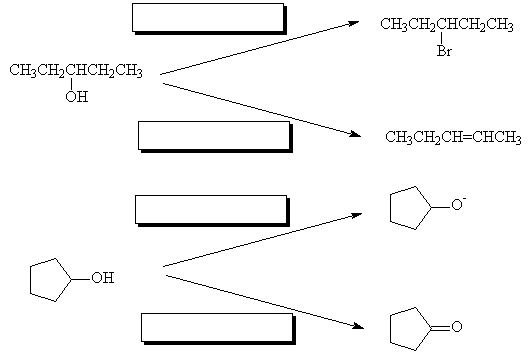
- What reagents are used in the following reactions?
- Draw curly arrows to describe the nucleophilic substitution reaction of 2-propanol with concentrated HI to form 2-iodopropane

- Draw the product formed in the following reactions:
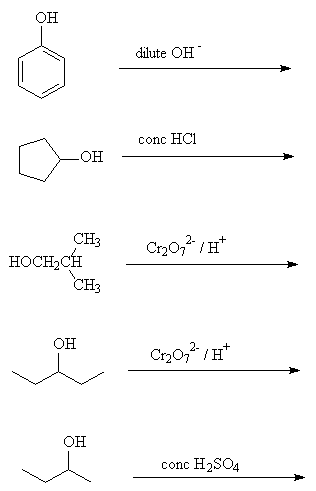
Go to answers
Return to Alcohols
Go to Index
ORGANIC HALOGEN COMPOUNDS
Predict the products from the following reactions:
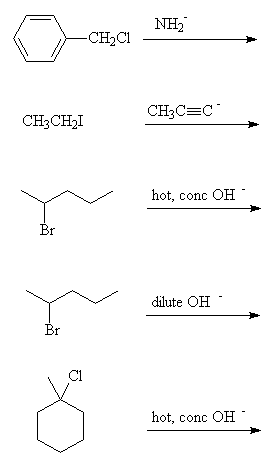
- Name and classify (1° , 2° , 3° or aryl) the following halogen compounds:

- Fill in the reagents in the following reaction scheme:
Go to answers
Return to Organic Halogen Compounds
Go to Index
ALDEHYDES AND KETONES
Name the following compounds

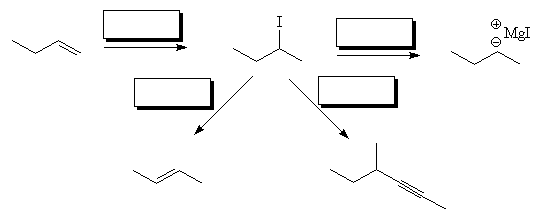
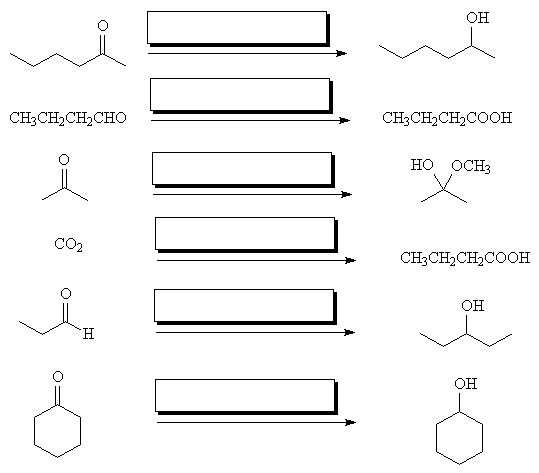
- Fill in the reagents in the following transformations:
- Use curly arrow notation to indicate the mechanism of nucleophilic addition of methyl magnesium bromide to acetone followed by treatment with aqueous acid.
- Draw the products of the following reactions:
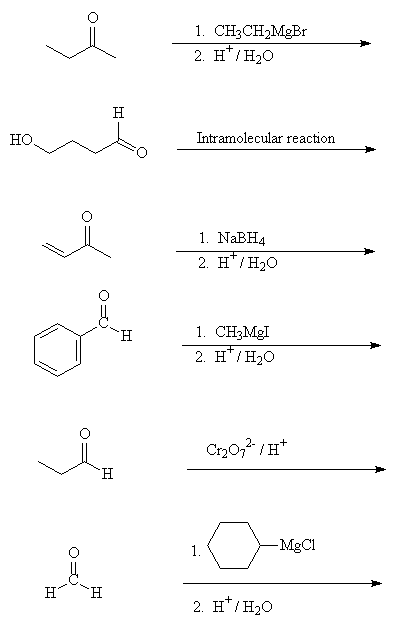
Go to answers
Return to Aldehydes and Ketones
Go to Index
CARBOXYLIC ACIDS
Name the following carboxylic acids

Go to answers
Return to Carboxylic Acids
Go to Index
CARBOXYLIC ACID DERIVATIVES
- Draw all the product(s) of the following reactions
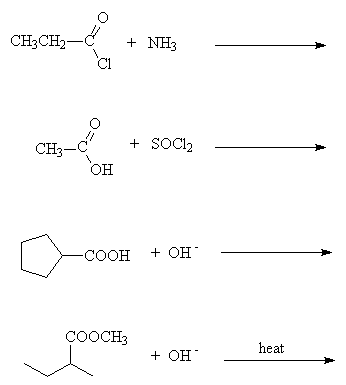
- Fill in the missing reagents
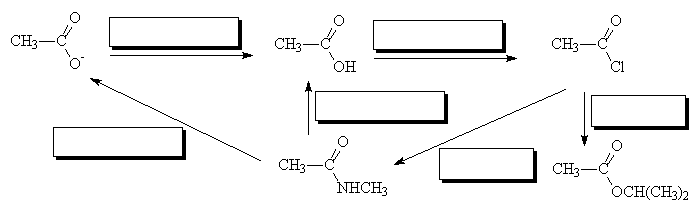
Go to answers
Return to Carboxylic Acid Derivatives
Go to Index
ISOMERISM AND STEREOCHEMISTRY
- Classify the following isomers as constitutional, conformational or diastereisomers:
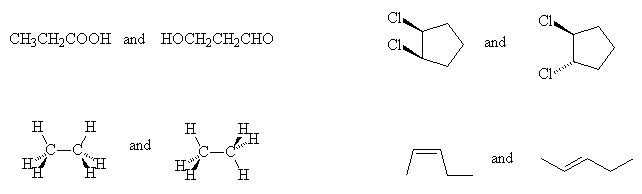
- Identify with an asterisk (*) the stereogenic carbon centres in the following molecules:
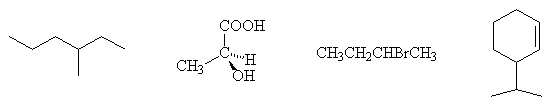
- Assign the configurations of the following stereogenic carbon centres:

- Identify the configuration (R or S) of the stereogenic carbon centre in each of the following compounds:
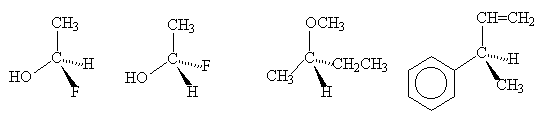
(i) (R)-2-methyl-3-pentanol
(ii) (S)-1-phenylethylamine
(iii) (2R,3S)-2,3-dihydroxybutane
- Which of the following compounds is/are meso-compounds ?
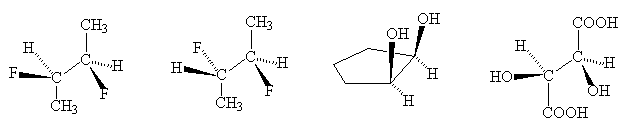
Go to answers
Return to Isomers and Stereochemistry
Go to Index