ISOMERISM AND STEREOCHEMISTRY
Reference: McMurry Ch 6 George et al Ch 3.3
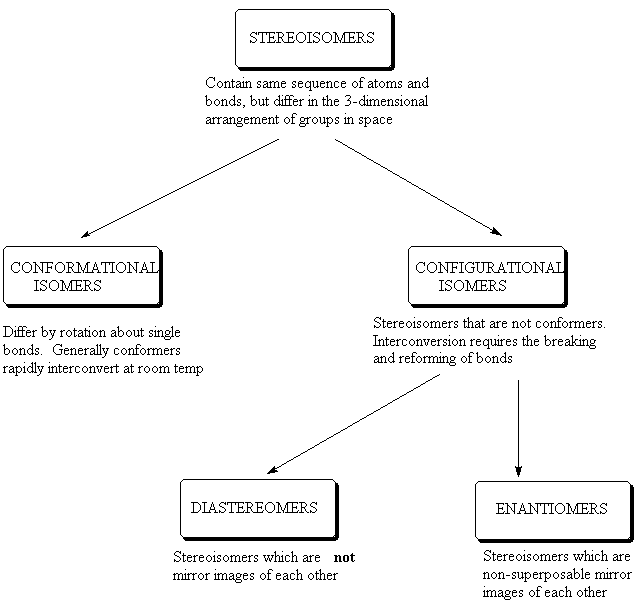
Recap: Some isomers have already been encountered – constitutional isomers (eg 1-butene and 2-butene); conformational isomers (e.g. staggered and eclipsed forms of ethane) and diastereomers such as (Z)- and (E)-2-butene or cis- and trans-1,2-dimethylcyclobutane.

Conformational isomers are one type of stereoisomer which generally can not be separated from one another at room temperature. Another class is configurational isomers, which can be separated from one another, as interconversion requires breaking of bonds. There are two types of configurational isomers: diastereisomers and enantiomers. Enantiomers are non-superposable mirror images.
Structures of Chiral Compounds
Example:
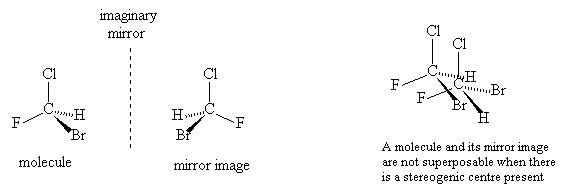
In molecules where there are not four different groups attached to a single carbon centre then any mirror image is identical to the original, just drawn in a different way.
Example:
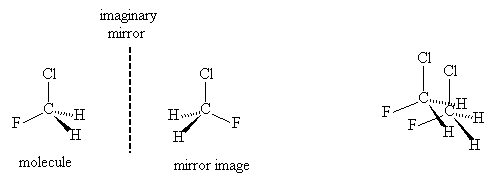
Enantiomers differ only in (i) the way that they interact with other chiral molecules, (ii) the way that they interact with polarised light.
NOMENCLATURE
The absolute configuration of a stereogenic centre refers to the exact three-dimensional arrangement of the groups. The method for naming enantiomers is called the Cahn-Ingold-Prelog convention and is based on describing the relative positions of the substituents attached to a stereogenic centre. Each stereogenic carbon centre has four different groups attached and these must firstly be assigned a relative priority according to a set of priority rules. These are the same rules used when naming (Z) and (E) alkenes.
The priority rules
(i) Each of the atoms attached directly to the stereogenic centre is assigned a priority based on its atomic number : the higher the atomic number, the higher the priority.
e.g. Increasing order of priority: H < C < N < O < Cl < Br < I
(ii) If two of the atoms attached to the stereogenic centre are identical (e.g. –CH3, -CH2CH3), then look to the next atoms away from the stereogenic centre. If the atoms at this point are still the same, then move further from the stereogenic centre (and so on) until the point of difference is found (there must be a point of difference somewhere for the molecule to be chiral). The group priority is then assigned based on the atomic numbers of the atoms at the first point of difference.
e.g. -CH3 < -CH2CH3 < -CH(CH3)2 < -CH2Cl < -CH2Br
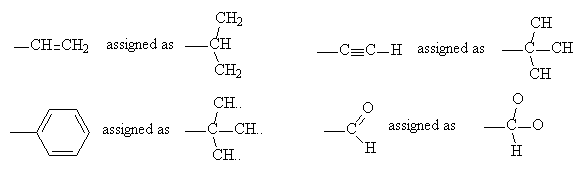
(iii) For the purposes of assigning group priorities, multiple bonds are expanded to be an equivalent number of single bonds attached to similar atoms. So a -CH=CH2 group is considered as equivalent to -CH(-CH2)2
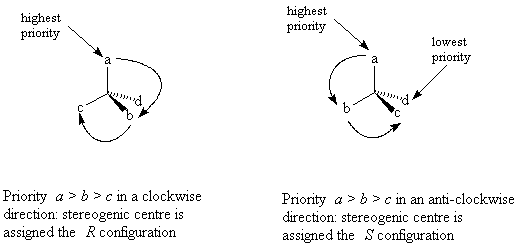
Assigning configuration
Rotate the molecule so the group of lowest priority is away from you. If going from the group of 1st to 2nd to 3rd priority traces a clockwise direction, the stereogenic centre is assigned the R configuration. If the path traced out is an anticlockwise direction, the stereogenic centre is assigned the S configuration.
Samples of Chiral Compounds
Chirality and chemical reactions
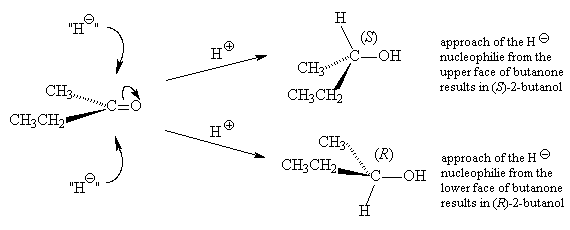
If the reaction begins with achiral starting materials and generates a compound that contains a stereogenic centre, then equal numbers of molecules with R- and S- configurations at that centre will be formed and the product must be a racemic mixture.
Example: Treatment of 2-butanone with 1) NaBH4 and 2) H+ /H2O forms a racemic mixture or 2-butanol.
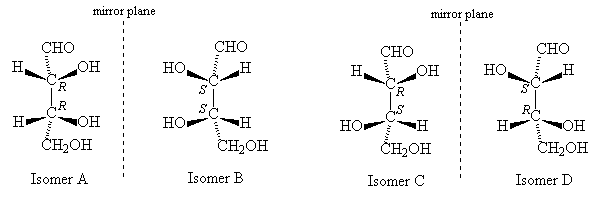
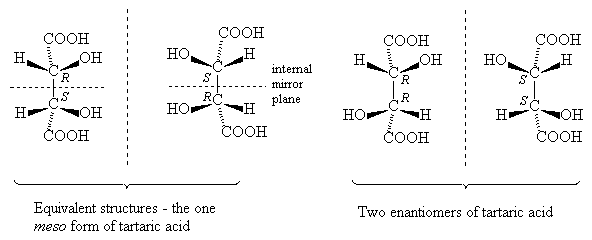
Molecules with more than one stereogenic centre
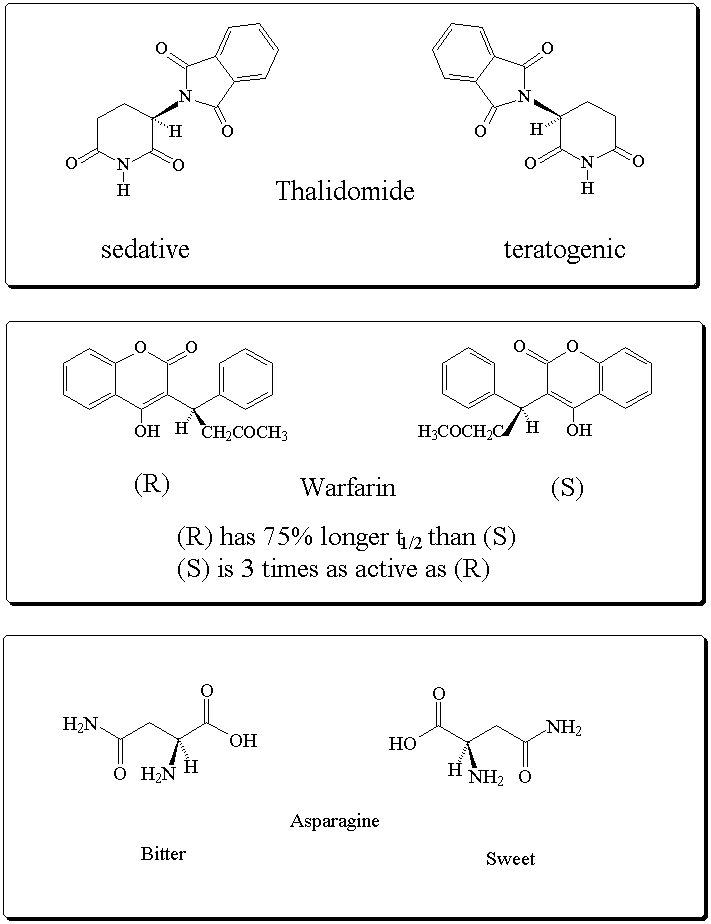
Some biologically active enantiomeric compounds: