Our research
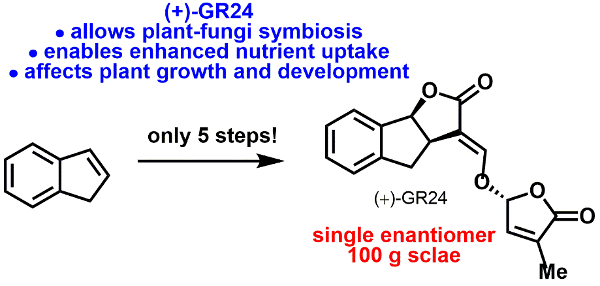
Strigolactone chemistry: Food security is an increasingly important issue. To increase crop-yields, one branch of current agrichemical research is centred on the development of plant growth promoters. We have a research program to generate the naturally occurring phytohormones called strigolactones, which hold great potential in this endeavour. We use a range of catalytic techniques to generate these molecules as single enantiomers.
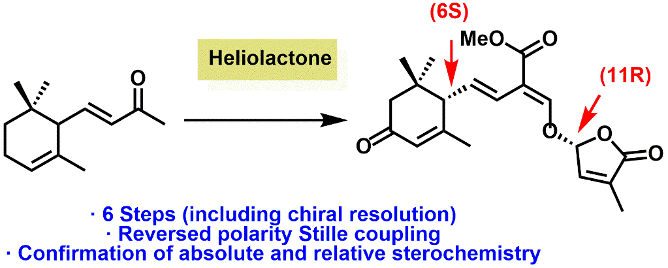
Additionally, we target strigolactones analogues and mimics.
Radical chemistry: As the smallest reactive unit, unpaired electrons (radicals) allow some bond-forming reactions to occur that would otherwise be difficult.
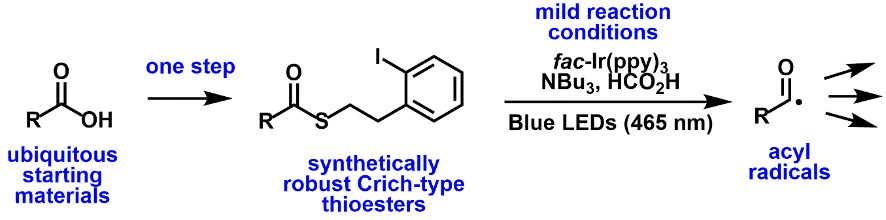
We have a program to generate radicals in a chemoselective and energy efficient manner for the streamlined synthesis of important targets.
Expanding the scope of the Stetter reaction:
The 'umpolung' addition of aldehydes onto unsaturated carbonyl compounds is an under-utilised method for carbon-carbon bond construction. We are investigating variants of this reaction that allow for the rapid construction of fused-ring systems from simple starting materials. We are also exploring the ability of the Stetter reaction to install new stereocentres under substrate control.
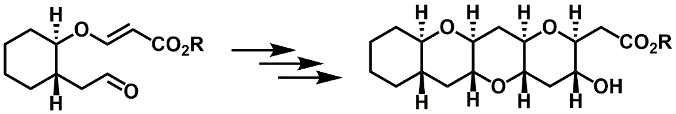
Fused cyclic ether synthesis: The classical Stetter reaction can be used to generated the trans, syn, transstereochemical arrangement seen in naturally occurring polycyclic ethers.
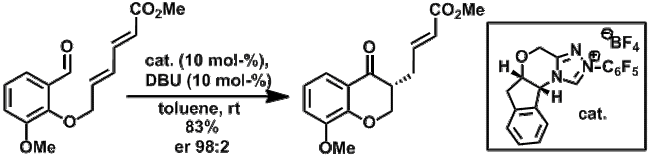
Stetter reactions with 1,6-acceptors: by using 'extended' Michael acceptors in the Stetter reaction, products with increased synthetic potential can easily be generated. The reaction can be conducted in an asymmetric fashion.
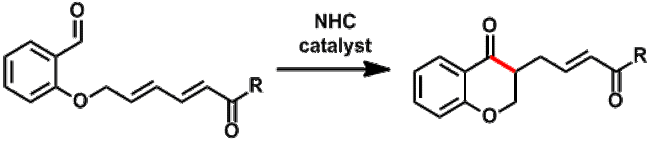
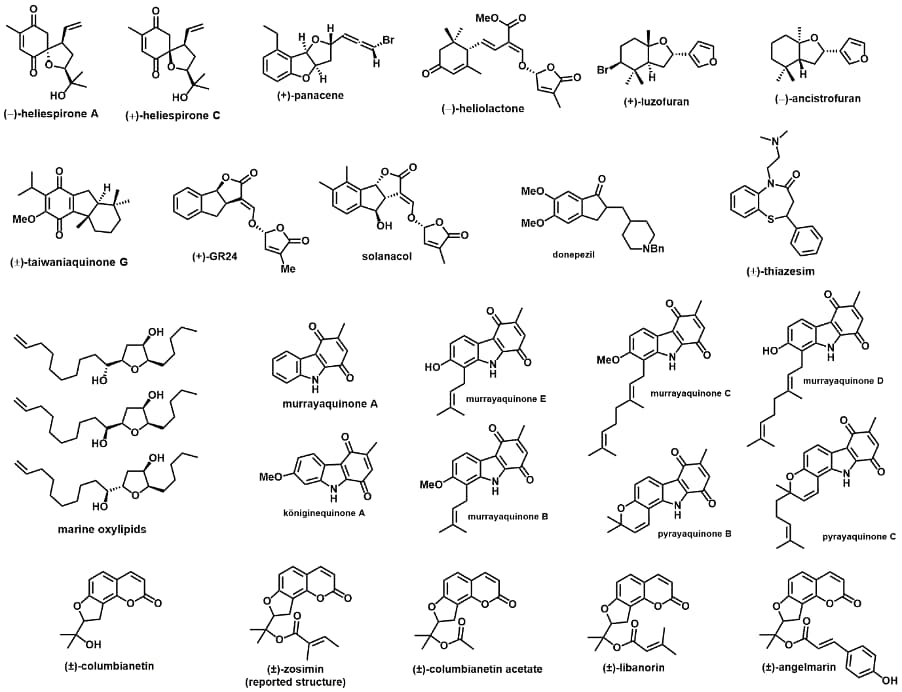
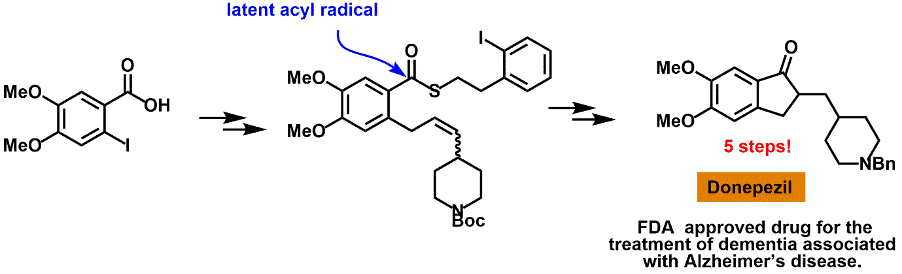
Completed targets: Below are some of the biologically relevant and structurally intriguing molecules that we have synthesised.